**Please note this article is part of a larger series that was first released in the June 2021 Monthly Playbook.
Please note this article is part of a larger series that was first released in the April 2021 Monthly Playbook. If you would like to receive the complete monthly playbook please join below.
If you would like to join the 1,512 others who are receiving high-signal, data-driven analysis to be in front of their peers in the cannabinoid space you can do so below. Each month we spend hours analyzing market research, data trends and private conservations to will keep you in front of the ever-evolving cannabinoid industry.
Cannabis Manufacturing For The 21st Century
Part 6: Overview of Extraction Methodologies & Monitoring
The separation of the wide array of compounds in cannabis sativa L. is becoming more important as regulations on cannabinoid containing products develop and become more stringent. There are a few common solvent methodologies used for extraction in the cannabis industry.
Alcohol extraction uses ethanol and is one of the most common and efficient methods of extracting the cannabinoids. The process can be done in either warm or cold ethanol. In warm or room temperature ethanol, the cannabinoids dissolve quickly leading to a high yield of cannabinoids. For warm ethanol extraction, the initial step is to soak the raw material in Ethanol to pull off the trichomes. In cold ethanol extraction, the first step is to chill the ethanol down to -40˚C before introducing the cold ethanol to the cannabis biomass.
Afterwards, the mixture is collected, filtered and the solvent is evaporated and reclaimed. In warm ethanol extractions, additional purification steps, such as winterization, are necessary. The biggest issue with scaling up this particular process is the ethanol itself. Because of its flammability, it is rated as a Class 1 Division 2 solvent, which limits the amount of Ethanol that can be stored on a particular site.[1] The evaporation of the solvent from the enriched liquid also requires specialized equipment to efficiently evaporate the ethanol to be reused in a closed-loop process.
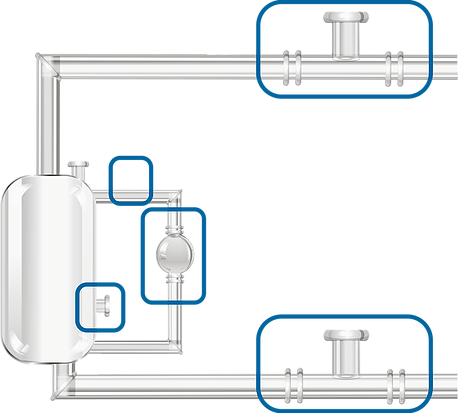
Hydrocarbon extraction generally uses either propane or butane. Similar to the cold alcohol method, the hydrocarbon solvent is cooled down to below it’s boiling point so that it is in liquid form. The initial extraction washes the raw material with the cold hydrocarbon solvent. The hydrocarbons with the products of interest can be separated by flowing the mixture into a separate area and raising the temperature. [2] With low boiling points, the hydrocarbons evaporate well below the point of degradation of some of the more volatile compounds, leaving behind the cannabinoids, terpenes, flavonoids and waxes.
After the separation of the hydrocarbons from the extract, the propane or butane can be recirculated though the biomass creating a closed-looped system. The result is an extract that is relatively free of inactive plant matter such as chlorophyll, fats and lipids. Inline de-waxing can also be accomplished using the same hydrocarbon solvent thereby entirely removing the need for winterization.
Overview of Extraction Methodologies & Monitoring

Despite this advantage, hydrocarbon extraction is losing popularity for several reasons including, but not limited to, regulations for handling propane/butane and stigma attached to a using a hazardous chemical for the extraction. [3] Similar to alcohol extraction, hydrocarbons are flammable, but they are more stringently classified as Class 1 Division 1. Another difficulty is that many places are banning the use of hydrocarbon extraction, making the setup and scale-up of this process unachievable.
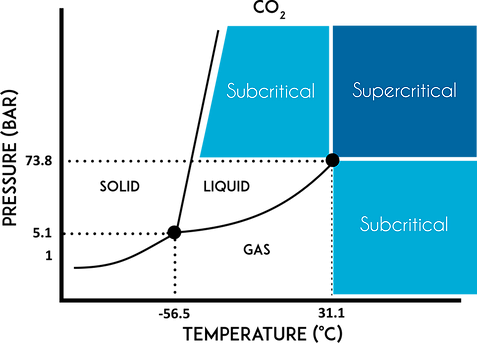
In the next issue, we’ll discuss the final extraction method: supercritical CO2. It’s many advantages make it a promising option for those looking to dive into extraction.